- Division of Antitumor Pharmacology, State Key Laboratory of Drug Research, Shanghai Institute of Materia Medica, Chinese Academy of Sciences, Shanghai 201203, China
- Department of Medicinal Chemistry, State Key Laboratory of Drug Research, Shanghai Institute of Materia Medica, Chinese Academy of Sciences, Shanghai 201203, China
- Department of Medical Oncology, Cancer Hospital, Shanghai Medical School, Fudan University, Shanghai 200032, China
- Department of Pharmacology, Shenyang Pharmaceutical University, 103 Wenhua Road, Shenhe District, Shenyang 110016, China
Molecular Cancer 2010, 9:268 doi:10.1186/1476-4598-9-268
The electronic version of this article is the complete one and can be found online at: http://www.molecular-cancer.com/content/9/1/268
Received: 26 January 2010
Accepted: 11 October 2010
Published: 11 October 2010
© 2010 Zhou et al; licensee BioMed Central Ltd.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Hypoxia-inducible factor-1α (HIF-1α), a critical transcription factor to reduced O2 availability, has been demonstrated to be extensively involved in tumor survival, aggressive progression, drug resistance and angiogenesis. Thus it has been considered as a potential anticancer target. Triptolide is the main principle responsible for the biological activities of the Traditional Chinese Medicine tripterygium wilfordii Hook F. Triptolide possesses great chemotherapy potential for cancer with its broad-spectrum anticancer, antiangiogenesis, and drug-resistance circumvention activities. Numerous biological molecules inhibited by triptolide have been viewed as its possible targets. However, the anticancer action mechanisms of triptolide remains to be further investigated. Here we used human ovarian SKOV-3 cancer cells as a model to probe the effect of triptolide on HIF-1α.
Triptolide was observed to inhibit the proliferation of SKOV-3 cells, and meanwhile, to enhance the accumulation of HIF-1α protein in SKOV-3, A549 and DU145 cells under different conditions. Triptolide did not change the kinetics or nuclear localization of HIF-1α protein or the 26 S proteasome activity in SKOV-3 cells. However, triptolide was found to increase the levels of HIF-1α mRNA. Unexpectedly, the HIF-1α protein induced by triptolide appeared to lose its transcriptional activity, as evidenced by the decreased mRNA levels of its target genes including VEGF, BNIP3 and CAIX. The results were further strengthened by the lowered secretion of VEGF protein, the reduced sprout outgrowth from the rat aorta rings and the inhibitory expression of the hypoxia responsive element-driven luciferase reporter gene. Moreover, the silencing of HIF-1α partially prevented the cytotoxicity and apoptosis triggered by triptolide.
The potent induction of HIF-1α protein involved in its cytotoxicity, together with the suppression of HIF-1 transcriptional activity, indicates the great therapeutic potential of triptolide as an anticancer drug. Meanwhile, our data further stress the possibility that HIF-1α functions in an unresolved nature or condition.
Hypoxia-inducible factor-1α (HIF-1α) is a critical transcription factor responsible for adaptive responses of cancer cells to reduced O2 availability(1). Through modulation of the expression of at least 70 genes, HIF-1α is extensively involved in tumor survival, aggressive progression, drug resistance and angiogenesis(2). Elevated levels of HIF-1α protein are observed in various human primary and metastatic cancers, either as direct results of intratumoral hypoxia or secondary to genetic alterations in oncogenes or tumor suppressor genes(3)(4). Those cancers are generally relatively poorly responsive to chemotherapy or radiotherapy with poor prognosis(5). Thus, HIF-1α has been proposed as a promising anticancer target(6). On the other hand, HIF-1α(-/-) tumors have also been demonstrated to show a feature of faster proliferation and more obvious resistance to apoptosis than the HIF-1α(+/+) counterparts(7), suggesting a possibility that HIF-1α may have unknown function(s) or exert its transcriptional activity dependent on specific, undefined conditions or stimulations.
Triptolide is an effective principle of the Traditional Chinese Medicine tripterygium wilfordii Hook F that has been used to treat autoimmune and inflammatory diseases for centuries(8)(9). Triptolide possesses broad-spectrum anticancer, antiangiogenesis, and drug-resistance circumvention activities(10)(11)(12)(13). Moreover, our recent study shows that a novel C14-hydroxyl substituted triptolide derivative elicits selective anticancer effects, specifically against ovarian and prostate cancers in nude mice xenograft models, with reduced toxicity as compared to the parent triptolide(14). Nevertheless, the anticancer action mechanisms of triptolide are complicated and remain to be further investigated. Triptolide downregulates various proteins including heat shock protein 70, Bcr-Abl, survivin, Mcl-1, Akt, c-myc, cyclin A/cdk2, cyclin B/cdc2, cyclin D1 and pRB, which may contribute to its anticancer activity under specific conditions(12)(15)(16). In addition, inhibition of nuclear factor κB activation by triptolide is also assoctiated with its potentiation of TNF related apoptosis-inducing ligand-induced anticancer effects(17). Notably, however, triptolide enhances the levels of p53 protein in p53-wild type human tumors but lowers its transcriptional activity, resulting in the reduced expression of p21 protein(18)(19)(20).
Based on the discovery of selective anticancer activity of the new triptolide analogue in human ovarian and prostate cancer xenograft models(14), in this current study, we used human ovarian SKOV-3 cancer cells as a model to further investigate the mechanisms of action of triptolide. Triptolide was unexpectedly revealed to enhance the cellular accumulation of HIF-1α protein with reduced transcriptional activity. The impact of triptolide on HIF-1α contributes to its partial anticancer effect. These results from triptolide further stress the possibility that HIF-1α functions in an unresolved nature or condition.
As expected, triptolide elicited potent concentration-dependent cytotoxicity in SKOV-3 cells with an averaged IC50 value of 10.24 nM for 72-h treatments (Fig. 1A). Unexpectedly, however, triptolide led to significant accumulation of cellular HIF-1α protein in a concentration-dependent manner following the 12-h exposure of SKOV-3 cells at normoxia (Fig. 1B). The similar results were reproducible in SKOV-3 cells exposed to hypoxia or mimic hypoxia with cobalt chloride (CoCl2) [23] (Fig. 1B) or in hypoxic human lung A549 and prostate DU145 cancer cells (Fig. 1C and 1D). The data indicate that the increase in the cellular accumulation of HIF-1α protein by triptolide is independent of the environmental oxygen pressure and the cell type, suggesting that it is an inherent capability of this agent.
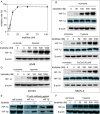
A. SKOV-3 cells were exposed to triptolide of gradient concentrations for 72 h. The proliferation inhibition was determined by SRB assays. B. SKOV-3 cells at normoxia (upper panel), 1% O2 hypoxia (middle panel) or CoCl2 (150 μM) mimic hypoxia (lower panel) were treated with tiptolide for 12 h and then subjected to standard Western blotting analyses for the levels of HIF-1α and HIF-1β proteins. C and D. A549 (C) and DU145 (D) cells at 1% O2 hypoxia were treated with tiptolide for 12 h and Western blotting analyses were done as in B. E. SKOV-3 cells at CoCl2 (150 μM) mimic hypoxia were treated with tiptolide at 1000 nM for 12 h. Then the cells were subjected to Western blotting for the levels of HIF-1α, HIF-1β proteins (left panel); or the cells were used to do co-immunoprecipitation assays for the binding between HIF-1α and HIF-1β (middle and right panels). F. SKOV-3 cells at 1% O2 hypoxia were treated with tiptolide at gradient concentrations for 12 h. Then the cells were subjected to Western blotting for the levels of HIF-2α proteins. All the experiments were performed three times and the representative results were presented.
Click image to view larger.
To function as a transcription factor, HIF-1α heterodimerizes HIF-1β that is constitutively expressed(2). The treatment with triptolide did not change the levels of HIF-1β in normoxic, hypoxic or CoCl2-treated SKOV-3 cells (Fig. 1B). Moreover, co-immunoprecipitation further showed that triptolide did not impair the binding of HIF-1α to HIF-1β (Fig. 1E). In addition, triptolide just marginally changed the levels of HIF-2α (Fig. 1F), another hypoxia-inducible factor (HIF) alpha subunit that has various overlapped targeting genes with HIF-1α(24).
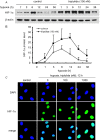
A and B. SKOV-3 cells were exposed to 1% O2 hypoxia in the presence or absence of triptolide (100 nM) for the indicated time periods. Then standard western blotting analyses were performed for the levels of HIF-1α protein (A). Adobe Photoshop CS2 version 9.0.2 was used for relative semi-quantification of the HIF-1α band intensity, which was normalized with β-Actin as the internal control. The results from three separate experiments were expressed as mean ± SD (B). C. SKOV-3 cells were treated as described in the Materials and Methods section and then subjected to immunofluorescence analyses for the localization of HIF-1α protein. The representative images were from three separate experiments with similar results.
Click image to view larger.

A. SKOV-3 cells were treated with triptolide for 12 h under hypoxia condition and then harvested for the detection of 26 S proteasome activity as described in the Materials and Methods. B. The lysates of SKOV-3 cells were incubated in the presence or absence of triptolide and then assayed for the 26 S proteasome activity. C and D. The levels of HIF-1α mRNA from reverse transcription -PCR (C) and real-time PCR (D) analyses in the hypoxic SKOV-3 cells treated with triptolide for 12 h. The levels of HIF-1α mRNA were normalized with β-actin mRNA expression; columns, mean of three independent experiments; bars, SD. The significant difference between triptolide-treated groups and hypoxia-control groups was analyzed by Student t test. * P < 0.05.
Click image to view larger.
To further dissect the possible cause of HIF-1α induction by triptolide, we used semi-quantitative RT-PCR and quantitative real-time PCR assays for the levels of HIF-1α mRNA in the triptolide-treated SKOV-3 cells. The results showed that triptolide enhanced the levels of HIF-1α mRNA in a concentration-dependent manner (Fig. 3C and 3D), which may be responsible for the HIF-1α accumulation.
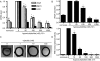
A. SKOV-3 cells were cultured under normoxic or hypoxic conditions in the presence or absence of triptolide for 12 h. The mRNA levels of VEGF, BNIP3 and CAIX were analyzed by real-time PCR and normalized with β-actin mRNA expression. B. ELISA assays were done for VEGF secretion from the SKOV-3 cells treated as in A. C. Triptolide inhibited new microvessel outgrowth arising from rat aorta rings. The representative images were from three separate experiments with similar results. D. MCF-7 cells were transiently transfected with the HRE-luciferase and renilla-luciferase reporter plasmids and then cultured at normoxia or hypoxia in the presence or absence of triptolide for 12 h followed by assays for luciferase activity. Data shown in A, B and D were expressed as mean ± SD, n = 3. The significant difference between triptolide-treated groups and hypoxia-control groups was analyzed by Student t test. * P < 0.05.
Click image to view larger.
To confirm whether triptolide reduces the transcriptional activity of HIF-1α protein, we used the hypoxia responsive element (HRE)-driven luciferase reporter gene assays. After failure with SKOV-3 or A549 cells due to the low transfection efficiency, we co-transfected the HRE luciferase reporter plasmid and the renilla luciferase reporter vector pGL-3 into MCF-7 cells for 24 h. Then the cells were treated with triptolide for additional 12 h. Triptolide reduced the luciferase-elicited fluorescence in a concentration-dependent fashion, and at 1000 nM of triptolide, the fluorescence almost lowered to the basal level (Fig. 4D). Collectively, the above evidence arising from all the levels of the transcription of the target genes, the reporter gene and the subsequent biological effects indicates that triptolide, though increasing the cellular accumulation of HIF-1α protein, reduces its transcriptional function.
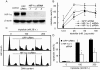
A. siRNAs targeting three different sequences of the HIF-1α gene were used to effectively silence this gene in SKOV-3 cells (Western blotting). GFP siRNA was used as scrambled control. B. The SKOV-3 cells transfected with HIF-1α or GFP siRNA were treated with triptolide for 48 h and then subjected to SRB assays for the proliferation inhibition. C. The SKOV-3 cells transfected with HIF-1α or GFP siRNA were treated with triptolide for 36 h and then assayed for apoptotic induction by flow cytometry as described in the Materials and Methods section. The representative histograms were from three independent experiments with similar results. D. The apoptosis rate from C. Data shown in B and D were expressed as mean ± SD, n = 3. The significant difference between triptolide-treated groups and hypoxia-control groups was analyzed by Student t test. * P < 0.05.
Click image to view larger.
Overexpression of HIF-1α in human cancers is usually correlated with poor prognosis and poor therapeutic response(32). HIF-1α has been considered as a potential therapeutic target(33). Nevertheless, there are also reports that HIF-1α null tumors grow unexpectedly fast, and that tumor cells with HIF-1α(-/-) tumors are more proliferative and less apoptotic than those with HIF-1α(+/+) even when tumor vessel formation was impaired(7). These paradoxical observations seem to suggest that the roles of HIF-1α in tumor development and/or in cancer therapy are conditional and that the conditions remain to be further clarified so that this important tumor-related factor can be better employed. Our findings in this current study further stress such conditional or stimulation-dependnent changes as the potent anticancer agent triptolide has been revealed to increase the cellular accumulation of HIF-1α protein that contributes partially to its anticancer activity.
Triptolide has been well documented to possess potent, broad-spectrum antitumor activity and to inhibit angiogenesis(10)(34). Our present study showed that triptolide enhanced the cellular accumulation of HIF-1α protein in SKOV-3, A549 and DU145 cancer cells at normoxia, hypoxia or CoCl2-mimic hypoxia. However, triptolide did not apparently change the protein levels of HIF-1β and HIF-2α and the binding of HIF-1α to HIF-1β. Triptolide did not change the kinetics or localization of HIF-1α protein in SKOV-3 cells exposed to hypoxia. Consistently, triptolide did not impair the 26 S proteasome activity. The increased levels of HIF-1α mRNA could be responsible for the accumulation of HIF-1α protein induced by triptolide in SKOV-3 cells. Unexpectedly, the triptolide-increased HIF-1α protein seems to lose its transcriptional activity, as evidenced by the decreased mRNA levels of its target genes including VEGF, BNIP3 and CAIX (the transcription of which is generally promoted by HIF-1α (30)(31)). The results were further strengthened by the lowered secretion of VEGF protein, the reduced sprout outgrowth from the rat aorta rings and the inhibitory expression of the HRE luciferase reporter gene. More importantly, the partial prevention of cytotoxicity and apoptotic induction of triptolide by HIF-1α silencing indicates the contribution of the accumulated HIF-1α protein to the anticancer action of this compound.
Several important points arise from those results: (1) The apparent association of the increased HIF-1α protein induced by triptolide with its anticancer activity challenges the general notion that the downregulation of HIF-1α protein elicits anticancer and antiangiogenic effects, as many HIF-1α inhibitors do(35)(36). (2) The HIF-1α protein in the triptolide-treated cells appears to have some additional non-transcriptional function because it loses its transcriptional activity but is indeed involved in the anticancer and antiangiogenic actvity of triptolide in our case. Another possibility is that the apparently decreased transcriptional activity of the triptolide-induced HIF-1α protein may be subsequent to the inhibitory effect of triptolide on RNA polymerase II (RNA Pol II). As a critical general transcription factor, RNA Pol II has been demonstrated to be inhibited by triptolide, which could contribute to its global transcriptional arrest(37)(38). The inhibition of RNA Pol II impairs the transcription-promoting function of HIF-1, thus reducing the transcriptional activity of the triptolide-induced HIF-1α protein. Actually, triptolide has been reported to similarly lead to the elevated levels of wild-type p53 protein with reduced transcriptional activity(18)(19)(20). (3) The enhancement of HIF-1α mRNA by triptolide may be a compensatory response by the tumor cells in an effort to maintain HIF-1α transcriptional activity. Or, subsequent to its RNA Pol II inhibition, the transcription of some factor(s) responsible for degrading HIF-1α mRNA may be arrested, finally disrupting the degradation machinery and resulting in the accumulation of HIF-1α mRNA. This is potentially similar to the effect of actinomycin D, an inhibitor of transcription, on the degradation of HIF-1α protein. Actinomycin D was found to lead to super-induction of HIF-1α protein by inhibiting the transcription of some unidentified factor(s) responsible for degrading HIF-1α protein (transcription-dependent degradation)(39). (4) Triptolide could be used as a tool compound to probe the potential new function of HIF-1α protein due to its unique action. (5) The interference of triptolide with HIF-1α is one of its possible anticancer and/or antiangiogenesis mechanisms.
Those points obviously deserve further investigation. Hypoxia is prevalent in solid tumors and HIF-1α is a critical regulator of tumor hypoxia adaption(40). Clarification of the conditions or stimulations required for specific HIF-1α activities (for instance, promoting or suppressing the expression of specific genes or gene sets) may greatly help the development of HIF-1α-targeted therapeutics and the monitor of cancer progression and prognosis. Moreover, demonstration of the function(s) of HIF-1α in addition to its transcriptional activity, if any indeed as shown in this study, will give new insights into hypoxia biology. On the other hand, triptolide is a promising anticancer lead compound and its chemical modifications are actively ongoing(14)(41). The discovery of its unique impact on HIF-1α suggests another possible anticancer mechanism. Therefore, the questions of how triptolide increases the levels of HIF-1α mRNA, why and how it reduces the transcriptional activity of HIF-1α protein need thoroughly elucidating.
In summary, triptolide was found to increase the levels of HIF-1α mRNA and protein and to reduce HIF-1α transcriptional activity, which contributes to its antitumor effect partially. These data, on one hand, propose a new potential anticancer mechanism of triptolide, and stress a possibility that HIF-1α functions in an unresolved nature or condition on the other.
The authors declare that they have no competing interests.
ZLZ, ZGL, JD and ZHM designed the study; ZLZ, ZGL, BY, YJ, YC, JMF, MD and LJT performed experiments; ZLZ, JD and ZHM analyzed data; ZL and YCL extracted and purified triptolide, tested its purity and wrote the chemical section. ZLZ and ZHM wrote the paper. All authors read and approved the final manuscript.
This study was supported by the grants from the National Natural Science Foundation of China (NSFC) (No.30721005 and No. 81025020), the National Science & Technology Major Project "Key New Drug Creation and Manufacturing Program" of China (No.2009ZX09301-001), the State Key Laboratory of Drug Research (No.SIMM0912QN-02) and National Basic Research Program of China (No 2010CB934000), respectively. We thank Dr. Giovanni Melillo of the Tumor Hypoxia Laboratory, National Cancer Institute at Frederick, USA for the kind gift of the HRE luciferase reporter plasmid. We also thank Mrs. Li-Juan Lu, Mr. Yong Xi and Miss Yan-Yan Shen for their technical supports.


