Author Affiliations
- Department of Surgical Sciences, Endocrine Unit, Uppsala University, SE-751 85 Uppsala, Sweden
- Department of Genetics and Pathology, Uppsala University, SE-751 85 Uppsala, Sweden
- Department of General Surgery, Martin-Luther-University of Halle-Wittenberg, 06097 Halle/Saale, Germany
Received: 2 June 2010
Accepted: 15 November 2010
Published: 15 November 2010
© 2010 Svedlund et al; licensee BioMed Central Ltd.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Parathyroid carcinoma (PC) is a very rare malignancy with a high tendency to recur locally, and recurrent disease is difficult to eradicate. In most western European countries and United States, these malignant neoplasms cause less than 1% of the cases with primary hyperparathyroidism, whereas incidence as high as 5% have been reported from Italy, Japan, and India. The molecular etiology of PC is poorly understood.
The APC (adenomatous polyposis coli) tumor suppressor gene was inactivated by DNA methylation in five analyzed PCs, as determined by RT-PCR, Western blotting, and quantitative bisulfite pyrosequencing analyses. This was accompanied by accumulation of stabilized active nonphosphorylated β-catenin, strongly suggesting aberrant activation of the WNT/β-catenin signaling pathway in these tumors. Treatment of a primary PC cell culture with the DNA hypomethylating agent 5-aza-2'-deoxycytidine (decitabine, Dacogen(r)) induced APC expression, reduced active nonphosphorylated β-catenin, inhibited cell growth, and caused apoptosis.
Aberrant WNT/β-catenin signaling by lost expression and DNA methylation of APC, and accumulation of active nonphosphorylated β-catenin was observed in the analyzed PCs. We suggest that adjuvant epigenetic therapy should be considered as an additional option in the treatment of patients with recurrent or metastatic parathyroid carcinoma.
Parathyroid carcinoma (PC) is a rare cause of primary hyperparathyroidism with characteristic marked hypercalcemia and highly elevated parathyroid hormone levels. In most western European countries and United States, these malignant neoplasms cause less then 1% of the cases, whereas incidence as high as 5% have been reported from Italy, Japan, and India(1)(2)(3)(4). The incidence in the United States has more than doubled since 1988(5). PCs are slow-growing with a high tendency to recur locally (30%-50%), and recurrent disease is difficult to eradicate(6)(7)(8). An overall 5-year survival rate of 86% and a 10-year survival rate of 49% have been observed in a report that described 286 cases of PC in the United States(9).
Loss of heterozygocity analyses have shown a higher fractional allelic loss in PCs than in parathyroid adenomas, where frequently deleted regions included the PTEN, RB, HRAS, p53, MEN1, and HRPT2 genes(10)(11). Somatic MEN1 mutations were recently described in 3 of 23 (13%) PCs, suggesting a role in the development of these tumors(12). Overexpression of the cyclin D1 oncogene (CCND1) has been observed in 10 of 11 (91%) PC specimens(13). Carriers of the hyperparathyroidism-jaw tumor (HPT-JT) syndrome show a higher risk of developing PC; 10% to 15% of affected individuals present with malignant parathyroid tumors(14). Somatic mutations of HRPT2 (parafibromin), the gene responsible for HPT-JT syndrome(14), occur in PC with an estimated odds ratio of 45.2 (95% CI: 12.8-160.0%) as calculated from a compilation of six published studies. The HRPT2 mutation frequency in sporadic parathyroid adenoma was calculated to 1.8%(15).
Negative immunostaining for parafibromin has been suggested as a means to diagnose PC, if the rare commonly cystic adenomas of the HPT-JT syndrome are excluded(16)(17). Recently, it was suggested that this may have some limited validity and cannot replace HRPT2 mutation analysis(18). Positive immunostaining for PGP9.5 (UCHL1) as well as absent staining for the adenomatous polyposis coli (APC) tumor suppressor have been suggested recently as additional markers for parathyroid malignancy(19)(20).
Inactivation of APC by mutation with subsequent stabilization of β-catenin has been strongly implicated in the cause of approximately 80% of colorectal cancers(21)(22)(23)(24). APC function can also be impaired by reduced expression through hypermethylation of the APC 1A promoter, a phenomenon observed in several malignances(25)(26)(27)(28)(29).
In the present study, we show that absent or markedly reduced expression of APC in five analyzed PCs is caused by hypermethylation of the APC promoter 1A. We also show that this was accompanied by accumulation of stabilized active nonphosphorylated β-catenin, strongly suggesting aberrant activation of the WNT/β-catenin signaling pathway in these tumors. Treatment of a primary PC cell culture with the DNA hypomethylating agent 5-aza-2'-deoxycytidine induced APC expression, reduced active nonphosphorylated β-catenin, inhibited cell growth, and caused apoptosis. 5-aza-2'-deoxycytidine (decitabine; Dacogen; MGI Pharma, Bloomington, MN) was recently approved by the United States Food and Drug Administration for the treatment of myelodysplastic syndrome(30)(31). We suggest that epigenetic therapy should be considered as an additional option in the treatment of patients with recurrent or metastatic parathyroid carcinoma.
This file can be viewed with: Microsoft Word Viewer
Format: DOC Size: 45KB Download file

A, RT-PCR analysis of parathyroid carcinomas (PC1-5), parathyroid adenomas (PA1-3), and normal parathyroid tissue specimens (PN1-5). B, Western blotting analysis of APC.
Click image to view larger.

Parathyroid carcinomas (in red; n = 5) and normal parathyroid tissue specimens (in blue; n = 8). Methylation levels were determined for ten CpGs of the APC promoter 1A, in triplicates. See Additional file 1, Table S1 for all measurements.
Click image to view larger.
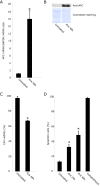
A, APC/GAPDH mRNA expression ratio as determined by real-time quantitative RT-PCR analysis. B, Western blotting analysis of APC. Coomassie blue-stained filter is shown as loading control. C, cell viability. D, apoptosis. Camptothecin at 0.1 μg/ml was used as positive control.
Click image to view larger.
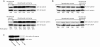
A, the five parathyroid carcinomas. Mean methylation levels (Additional file 1, Table S1) of the APC promoter 1A are also shown. B, normal parathyroid tissue specimens (n = 5). C, active β-catenin level in cultured primary PC1 cells treated with 5-aza-2'-deoxycytidine for the indicated time points.
Click image to view larger.
For all five analyzed PCs, our results strongly suggest inactivation of APC by DNA methylation and consequential aberrant WNT/β-catenin signaling by the accumulation of active nonphosphorylated β-catenin. Treatment in vitro with the hypomethylating agent 5-aza-2'-deoxycytidine inhibited PC cell growth and induced apoptosis, raising hopes for adjuvant therapy. A recent study demonstrated negative immunohistochemical staining for APC in nine of twelve PCs(20), supporting a frequent inactivation of APC in PCs.
The only treatment of PC with higher rate of cure is diagnosis before or during operation followed by complete removal of the tumor. Treatments of recurrent or metastatic disease have been difficult to evaluate due to the uncommonness of the tumor entity, but in one study adjuvant radiotherapy was shown to improve survival(43). 5-aza-2'-deoxycytidine (decitabine; Dacogen; MGI Pharma, Bloomington, MN) was recently approved by the United States Food and Drug Administration for the treatment of myelodysplastic syndrome(30)(31). 5-aza-2'-deoxycytidine has been shown to suppress tumorigenecity of xenografted cells from solid tumors(41)(44)(45)(46). Although its efficacy in solid tumors remains to be fully evaluated (31)(47), we suggest that Dacogen(r) should be taken under consideration for additional treatment of recurrent or metastatic PC, a life threatening condition which has been notoriously difficult to eradicate.
The WNT/β-catenin signaling pathway was aberrantly activated in the analyzed parathyroid carcinomas, due to lost expression of APC likely caused by promoter DNA methylation. APC was reactivated in vitro by 5-aza-2'-deoxycytidine and adjuvant epigenetic therapy (decitabine; Dacogen; MGI Pharma, Bloomington, MN) should be considered as an additional option in the treatment of patients with recurrent or metastatic parathyroid carcinoma.
The authors declare that they have no competing interests.
JS performed experiments, analyzed data, performed statistical analysis, and helped to draft the manuscript. MA performed experiments, MS analyzed data and helped to draft the manuscript. HD contributed with material and helped to draft the manuscript. GÅ helped to draft the manuscript. PB performed experiments, analyzed data, performed statistical analysis, and helped to draft the manuscript. GW conceived of the study, participated in its design and coordination and drafted the manuscript. All authors read and approved the final manuscript.
This work was supported by the Swedish Research Council, Swedish Cancer Society, and Lions Fund for Cancer Research.


