- *Corresponding author: Haiquan Chen hqchen1@yahoo.com
- † Equal contributors
- Center of Lung Cancer Prevention & Treatment, Department of Thoracic Surgery, Shanghai Cancer Hospital, Fudan University; Department of Oncology, Shanghai Medical College, Fudan University, Shanghai 200032, China
- Institute for Nutritional Sciences, Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences; Graduate School of the Chinese Academy of Sciences, 200031, China
| Received: | 5 January 2011 |
| Accepted: | 20 May 2011 |
| Published: | 20 May 2011 |
© 2011 Wang et al; licensee BioMed Central Ltd.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Monocyte recruited into the tumor and maturation to tumor-associated macrophage (TAM). Interleukin-10(IL-10) is a potent immunosuppressive cytokine, which can be secreted from both primary tumor and stromal cells. However, there are controversies regarding its role in the progression of cancer. So it is important to isolate TAM from tumor cells to study the role of IL-10 in the progress of cancer. The aim of our study was to determine whether IL-10 expressed by TAM correlated with clinicopathological factors in NSCLC.
TAM in NSCLC was isolated by short-term culture in serum free medium with the modification to literature reports. The mRNA expression levels of IL-10, cathepsin B, cathepsin S, which were closely related with TAM according to the literatures, were evaluated by Quantitative real-time RT-PCR in 63 NSCLC. The relationships between their expression levels and clinicopathological features were investigated.
We successfully achieved up to 95% purity of TAM, derived from 63 primary lung cancer tissues. TAM expressed high levels of IL-10, cathepsin B in NSCLC. High levels of IL-10 in TAM significantly correlated with stage, tumor size, lymph node metastasis, lymphovascular invasion or histologic poor differentiation.
Our results revealed that TAM with high levels of IL-10 expression may play an important role in the progression of non-small cell lung cancer. The data also suggested that TAMs may involve in tumor immunosuppression through overexpressed IL-10. Additionally, the phenotype of isolated TAM can be potentially used to predict clinicopathological features as well.
Lung cancer; Tumor associated macrophages; IL-10
Tumor-associated macrophages (TAMs) are the most abundant cancer stromal cells involved in the host immune system(1)(2). In recent years, increasing attention has focused on TAMs, unique macrophage populations that play pivotal roles in tumor immunosuppression, and provide a suitable microenvironment for cancer development and progression(3). TAM infiltration has been found to be correlated with a worse outcome in several malignant tumors(4)(5)(6)(7)(8)(9). The possible mechanism by which TAMs support tumor progression and help the tumor evade immunosurveillance is through the release a spectrum of tumor promoting and immunosuppressive products.
Interleukin-10(IL-10), cathepsin B or cathepsin S was reported to be closely associated with TAMs in recent literatures(10)(11)(12). IL-10 is produced primarily by T cells, B cells, dendritic cells, and monocytes/macrophages(13). Tumor-associated macrophages form a major component in a tumor, and have been suggested to play an essential role in the complex process of tumor-microenvironment coevolution and tumorigenesis(1). Previous reports have also shown that TAMs produce high levels of IL-10, exhibit little cytotoxicity for tumor cells(14). However, there are controversies regarding its role in the progression of cancer(15)(16). So it is important to isolate TAM from tumor cells to study the role of IL-10 in the progress of cancer.
By using DNA-microarray technology, recent study demonstrated that NSCLC patients with a high expression level of cathepsins in lung cancer tissue (both tumor cells and stroma cells) had a poor outcome(17). Interestingly, it has been shown that TAM is the primary source of high levels of cathepsin activity in pancreatic, breast and prostate cancer animal models(10)(11)(12). However, the significance of cathepsins expressed by TAM in NSCLC remains unknown.
In the present study, we assessed IL-10, cathepsin B and cathepsin S expression in TAMs, freshly isolated from lung tumor tissue, in correlation with clinicopathological factors in NSCLC.
63 paired peripheral blood samples and primary lung cancer tissues were collected from patients before or at the time of surgical resection at the Center for Lung Cancer Prevention and Treatment of Shanghai Cancer Hospital from June 2009 to March 2010. Data collected included age, sex, smoking history, histopathological diagnosis, TNM stage, lymphovascular invasion, pleural invasion, and tumor differentiation. Histological diagnoses, presence of lymphovascular invasion(LVI), and grade of differentiation were confirmed by two senior histopathologists. A consent form was signed by every patient or his/her legal representatives. This study was approved by the committees for Ethical Review of Research at Shanghai Cancer Hospital.
Histological diagnosis and grade of differentiation were determined in accordance with the World Health Organization criteria for lung cancer(18). The pathologic tumor stage (p stage) was determined according to the revised TNM classification of lung cancer(19).
TAMs were isolated from solid tumors according to literature reports(20)(21)(22). Briefly, Tumor tissue was cut into 2 mm fragments, followed by collagenase digestion (0.3 mg/ml, Worthington Biochemical Corp, NJ, USA) for 1 h at 37°C. The suspension was filtered through a 70 μm stainless steel wire mesh to generate a single-cell suspension. The suspension was centrifuged and washed twice with PBS. Cells were left to adhere in serum-free RPMI 1640 for 40 min. Nonadherent cells were washed away. Ninety-five percent of the remaining adherent cells were TAMs as assessed by morphology and macrophage specific marker CD68 positivity.
TAMs were adhered to 24-well plate , fixed in 4% paraformaldehyde at room temperature for 5 minutes, washed with PBS twice, incubated with 1% BSA at 37°C for 30 minutes to block nonspecific interactions, and then stained with primary antibodies to CD68 (1:100 dilution, sc-20060, Santa Cruz Biotechnology, CA, USA) at 4°C overnight. After several washes with PBS, the cells were incubated in an appropriate, rhodamine-labeled goat anti-mouse secondary antibody(Proteintech Group, Inc, Chicago ,USA) at room temperature for 1 h. Nuclei of all cells were then stained with 4'6-diamidino-2-phenylindole(DAPI). Image was taken at 200 × magnification on an Olympus-IX51 microscope. For each patient, 10 fields were imaged and measured for percentage of macrophage (CD68 positive cells/DAPI stained cells). Immunofluorescence was repeated in three randomly selected patients.
Macrophage (Mφ) was obtained as described previously(20). In brief, the mononuclear cells were isolated from peripheral blood matched with TAMs by Ficoll-Hypaque density gradient centrifugation (density, 1.077 ± 0.001 g/ml, Axis-Shield, Oslo, Norway) at 450 × g for 30 min at room temperature. The mononuclear cells were washed thrice with PBS and plated at 1 × 107 in 6-cm tissue culture dishe for 2 h in DMEM alone. Thereafter, the nonadherent cells were washed thrice with warm PBS and the adherent monocytes were cultured in DMEM containing 5% FBS and 25 ng/ml human macrophage colony-stimulating factor((rhM-CSF, PeproTech, Rocky Hill, NJ, USA), The medium was changed every 2 days, and macrophage were obtained after 6 days in vitro cultivation.
Total RNA was isolated from TAMs and their matched macrophages by using RNeasy Mini Kit (Qiagen, Valencia, CA, USA) as described by the manufacturer's protocol. For mRNA analysis, an aliquot containing 2 μg of total RNA was transcribed reversely using M-MLV reverse transcriptase (Promega, Madison, WI, USA). Specific primers (Genery, Shanghai, China) were used to amplify cDNA. QRT-PCR was done using SYBR Green PCR master mix (Applied Biosystems, Piscataway, NJ, USA). The primers for QRT-PCR were: β-actin forward (F) 5' ACCACA CCTTCTACAATGA3', β-actin reverse(R) 5'GTCATCTTCTCGCGGTTG3'; IL-10 F 5' AGAACCT GAAGACCCTCAGGC3', IL-10 R 5' CCACGGCCTTGCTCTTGTT 3'; cathepsin B F 5' TGCA GCGCTGGGTGGATCTA 3'; cathepsin B R 5' ATTGGCCAACACCAGCAGGC 3'; cathepsin S F 5' GCTTCTCTTGGT GTCCATAC 3', cathepsin S R 5' CATTACTGCGGGAATGAGAC 3'. The amplification protocol consisted of an initial 10 min denaturation step at 95°C, followed by 40 cycles of PCR at 95°C for 15s, 60°C for 1 min and detection by the ABI-Prism 7900HT Sequence Detection System (Applied Biosystems, Foster City, CA, USA). Each sample was assayed in triplicate. The comparative CT method (ΔΔCT method) was used to determine the quantity of the target sequences in TAM relative both to Mφ (calibrator) and to β-actin (an endogenous control). Relative expression levels were presented as the relative fold change and calculated using the formula: 2 -ΔΔCT= 2-(ΔCT (TAM) - ΔCT (Mφ) where each ΔCT =ΔCT target-ΔCT β-actin.
For exact identification of IL-10 or cathepsin B expression in TAMs, serial sections were used to examine the expression of IL-10, cathepsin B in TAMs. Samples were fixed in 4% formaldehyde in PBS (pH 7.2) and paraffin embedded. 4-μm thickness was cut from each paraffin block. After dewaxing and rehydration, the sections were microwaved for antigen retrieval in 10 mmol/liter citrate buffer (pH 6.0) for 10 min, and then allowed to cool for 1 hour at room temperature. Endogenous peroxidase activity was blocked with hydrogen peroxide; Nonspecific binding was blocked by preincubation with 10% goat serum in PBS for 30 minutes at room temperature. Slides were incubated with the primary antibodies directed against monoclonal anti-human CD68 antibody (1:200 dilution, sc-20060, Santa Cruz, CA, USA), monoclonal anti-human IL-10 antibody (1:100 dilution, BA1201,Boster, WuHan, China) or polyclonal anti-human cathepsin B antibody (1:100 dilution, ab49232, Abcam, MA, USA). The results were visualized using the streptavidine-biotin immunoperoxidase detection kit and AEC chromogen (Maixin Bio, FuZhou, China) based on the manufacturer's instruction. Positive cells stained red. The negative control involved omission of the primary antibody.
Statistical analysis software (Prism 5.0, GraphPad Software Inc, La Jolla, CA, USA and SPSS Version 13.0 software, SPSS Inc, Chicago, IL, USA) was used to perform the analyses. Data are expressed as median (range). The Mann-Whitney test was used for the comparison between TAM and normal macrophage. The correlation between IL-10 or cathepsin B expression and clinicopathologic factors was analyzed by Mann-Whitney test. Multivariate logistic regression was performed to evaluate the relationships between the pathological stage (with early and late stage as dependent variables) and covariates (age, sex, tobacco use, tumor histology and IL-10 expression in TAMs). For this analysis, the median value of IL-10 was chosen as the cut-off point for dividing the patients into the two groups. Two-tailed P value less than 0.05 was considered statistically significant.
The patient characteristics are described in Table 1. Patients (40 males and 23 females) had a mean age of 58.8 ± 1.1 years. Fifty-four patients had a smoking history, and forty-six were non-smokers. Adenocarcinoma was the most common tumor type (54%) followed by squamous cell carcinoma (32%). 30 patients (48%) were stage I (early stage), and the remaining 34 patients were (52%) stages II, III or IV (late stage) of the disease.
|
characteristic
|
No.
a
(N= 63)
|
%
|
|
|---|---|---|---|
| Age/years(Median,range) |
58 (37-76) |
||
| Sex |
|||
| Male |
40 |
63.5 |
|
| Female |
23 |
36.5 |
|
| Tobacco use |
|||
| Current |
22 |
35 |
|
| Former |
12 |
19 |
|
| Never |
29 |
46 |
|
| Histology |
|||
| Adenocarcinoma |
34 |
54 |
|
| Squamous cell carcinoma |
20 |
32 |
|
| Othersb |
9 |
14 |
|
| Stage |
|||
| StageⅠ |
30 |
48 |
|
| StageⅡ |
11 |
17 |
|
| StageⅢ |
17 |
27 |
|
| StageⅣc |
5 |
8 |
|
| Lymph node metastasis |
|||
| N0 |
42 |
67 |
|
| N1/N2 |
21 |
33 |
|
| Pleural invasion |
|||
| Negative |
43 |
68 |
|
| Positive |
20 |
32 |
|
| Lymphovascular invasion |
|||
| Negative |
51 |
81 |
|
| Positive |
12 |
9 |
|
| Histologic differentiation |
|||
| Well/Moderate |
30 |
48 |
|
| Poor |
26 |
41 |
|
| not availabled |
7 |
11 |
|
A Number for all except age.
B Include 2 Large cell carcinoma, 2 Carcinoid, 1 malignant clear cell sugar tumor, 1 Sarcomatoid carcinoma, and 3 malignancy, but type undetermined.
CStage IV was found incidentally during the operation or only for biopsy
D Include Carcinoid, malignant clear cell sugar tumor, Sarcomatoid carcinoma, multiple primary lung cancer.
Wang et al. Journal of Experimental & Clinical Cancer Research 2011 30:62 doi:10.1186/1756-9966-30-62
In our study, 71 NSCLC samples were collected and TAMs were successful isolated from all samples. However, cell number of TAMs isolated from 8 NSCLC was inadequate for gene expression analysis, and excluded from this study. So TAMs from 63 NSCLC were finally analyzed. The successful rate was 89%(63/71). Each sample weight ranged from 10 mg to 200 mg and the cell number of TAMs collected ranged from 5 × 105 to 1 × 107 per 100 mg tumor tissue.
TAMs from lung cancer tissue had an irregular shape and projections (Figure 1A). To confirm that the cell isolated from the lung cancer tissue were TAMs without contamination by fibroblasts or tumor cells, staining for the macrophage specific marker CD68 was performed. Over ninety-five percent of the cells stained positively for each randomly selected patient (Figure 1B).

A. Representative cell morphology of tumor-associated macrophages, TAM, fibroblast and lung tumor cell. B. Immunofluorescent was used to distinguish macrophage, fibroblast and lung tumor cell with antibodies targeting CD68 (red), nuclei stained with DAPI (blue). Original magnification, × 400.
Click image to view larger.
We performed a time course study to show the expression level of IL-10, cathepsin B and cathepsin S in monocytes changes after culture in medium with rhM-CSF. Our study showed the expression level of IL-10, cathepsin B and cathepsin S showed no significant changes in the time dependent study. (All p > 0.05) (Figure 2A). We also performed dose depedent study of rhM-CSF to see whether the expression level of IL-10, cathepsin B and cathepsin S were affected or not. Our study showed that the dose of rhM-CSF did not affect the expression level of IL-10, cathepsin B and cathepsin S (Figure 2B).
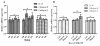
Results are given as fold increase in mRNA expression with respect to expression in D0 monocytes. Data were normalized to expression of the β-actin gene. A: Monocytes(D0) was used as a calibrator. B, monocytes culture without rhM-CSF was used as a calibrator (Ctrl). Error bar is SD, Independent experiments were repeated three times, all #p > 0.05(by student t-test).
Click image to view larger.
The mRNA expression levels of IL-10, cathepsin B and cathepsin S in TAMs were analyzed using QRT-PCR, compared with matched normal macrophages from the 63 patients. To explore the best time point for analyzing the expression level of IL-10, cathepsin B and cathepsin S, a time course study was done. After adhere to plastic for 20 min, 40 min, 60 min and 90 min, the expression level of IL-10 were: 28.3 ± 2.3; 28.1 ± 1.1; 24.6 ± 2.1; 14.7 ± 2.9 respectively, and the purity of TAMs were: 100%, 97%, 95%, 84% respectively (staining for the macrophage specific marker CD68 was performed). After 60 min, tumor cells and fibroblast began to adhere, the purity decreased rapidly. So we chose 40 min as the time point for adherence, which is consistent with previous reports(23)(Figure 3).
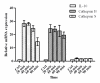
Results are given as fold increase in mRNA expression with respect to expression in ctrl (normal macrophages). Data were normalized to expression of the β-actin gene. Normal macrophages were used as a calibrator. Error bar is SD; Independent experiments were repeated three times.
Click image to view larger.
Compared with the expression in macrophage, IL-10 and cathepsin B were significantly upregulated (p < 0.05). After normalized to macrophages, the median values (range) of each gene in TAM were: IL-10, 30.5(0.6-530.3) and cathepsin B, 11.9(0.6-69.1) (Figure 4 A-B). There were no significant differences in the level of cathepsin S between the TAMs(0.85(0.04-4.49))and the macrophages (Figure 4C).
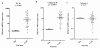
Results are given as fold increase in mRNA expression with respect to expression in matched Mφ. Data were normalized to expression of the β-actin gene. Mφ was used as a calibrator. Bars represent median. *p by the Mann-Whitney U test.
Click image to view larger.
To confirm whether TAMs express IL-10 and cathepsin B in protein level, 6 NSCLC (3 late stage (IIIA) and 3 early stage (Ia- Ib)) were randomly selected to perform IHC using antibody against CD68, IL-10 and cathepsin B on serial sections. We demonstrated that almost all CD68 positive cells co-expressing IL-10, which in line with the QRT-PCR results that the IL-10 mRNA expression level is high (Figure 5 A-B). The IL-10 expression was negative by IHC in 3 early stage NSCLC, which in line with the QRT-PCR results that the IL-10 mRNA expression level below the median (30.5) in 3 early stage NSCLC. Expression of cathepsin B in macrophage was observed in 5 of 6 cases. Among macrophages expressing cathepsin B, only a small portion of the cells showed strong positive (Figure 5 C-D) and not associated with stage of disease.
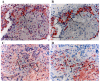
A-B, High IL-10 expression in macrophage, A, IL-10 staining in macrophage (strong positivity); B, CD68 staining. C-D, Cathepsin B expression in macrophage; C, cathepsin B staining in macrophage (most cells were moderate positivity, only a few cells were strong staining); D, CD68 staining. Scale bar indicates 50 μm. Original magnification, × 400.
Click image to view larger.
The correlation between IL-10, cathepsin B expression in TAM and clinicopathologic factors was shown in Table 2. A strongly positive correlation between IL-10 mRNA expression in TAM and tumor stage was seen. Increased expression levels of IL-10 in TAM were seen in NSCLC patients with late stage (stage II, III and IV). When multivariate logistic regression analysis was performed, IL-10 expression in TAMs was shown to be an independent predictive factor for late stage disease (Table 3).
|
IL-10
|
Cathepsin B
|
||||
|---|---|---|---|---|---|
|
Variables
|
N
|
Median(Range)
|
p* value
|
Median (Range)
|
p* value
|
| age |
|||||
| <58 |
26 |
31.3(3.05-530.3) |
0.252 |
10.9(0.9-51.9) |
0.41 |
| ≥58 |
37 |
30.5(0.6-511.6) |
14.5(0.6-69.1) |
||
| Gender |
|||||
| Male |
40 |
31.3(1.3-530.3) |
0.607 |
14.9(0.9-69.1) |
0.061 |
| Female |
23 |
19.9(0.6-426.1) |
10.1(0.6-37.9) |
||
| Smoking history |
|||||
| Never |
29 |
30.5(0.6-426.1) |
0.699 |
10.1(0.6-51.9) |
0.067 |
| Former or current |
34 |
31.2(1.3-530.3) |
14.9(1.5-69.1) |
||
| Histology |
|||||
| Adenocarcinoma |
34 |
42.9(0.6-530.3) |
0.045 |
12.7(0.6-69.1) |
0.41 |
| Squamous cell carcinoma |
20 |
17.1(1.3-354.3) |
16.6(1.5-41.7) |
||
| Others |
9 |
41.2(6.4-511.6) |
10.2(4.2-26.7) |
||
| Pathological stage |
|||||
| Stage I |
30 |
9.7(0.6-140.8) |
0.016 |
13.1(0.6-69.1) |
0.066 |
| StageⅡ |
11 |
28.9(1.8-511.6) |
13.6(3.1-41.7) |
||
| StageⅢ |
17 |
177.7(23.5-530.3) |
11.8(1.2-51.9) |
||
| StageⅣ |
5 |
249.9(55.4-429.9) |
10.1(3.6-25.9) |
||
| T status |
|||||
| T1 |
15 |
4.1(0.6-263.6) |
<0.0001 |
9.9(0.6-22.7) |
0.037 |
| T2-3 |
48 |
42.9(1.6-530.3) |
14.2(0.9-69.1) |
||
| Lymph node metastasis |
|||||
| N(+) |
21 |
119.1(6.1-530.3) |
<0.0001 |
13.6(1.2-46.9) |
0.466 |
| N(-) |
42 |
19.2(0.6-273.8) |
11.1(0.6-69.1) |
||
| Lymphovascular invasion |
|||||
| LVI(+) |
12 |
93.1(6.2-530.3) |
0.01 |
14.2(0.9-37.8) |
0.92 |
| LVI(-) |
51 |
26.5(0.6-429.9) |
11.1(0.6-69.1) |
||
| Pleural invasion |
|||||
| PL(+) |
20 |
55.8(14.9-530.3) |
0.002 |
14.2(0.9-69.1) |
0.376 |
| PL(-) |
43 |
19.9(0.6-354.9) |
11.1(0.6-51.9) |
||
| Differentiation |
|||||
| Well or Moderately |
30 |
17.3(0.6-429.9) |
0.001 |
13.0(0.6-69.1) |
0.961 |
| poorly |
26 |
113.1(1.6-530.3) |
11.9(1.2-37.9) |
||
*p by the Mann-Whitney U test
Wang et al. Journal of Experimental & Clinical Cancer Research 2011 30:62 doi:10.1186/1756-9966-30-62
|
B
|
SEM
|
Chi-squared
|
p-value
|
OR (95% CI)
|
|
|---|---|---|---|---|---|
|
Sex
|
0.241 |
1.110 |
0.037 |
0.847 |
1.239(0.141-10.922) |
|
Age
|
-0.063 |
0.040 |
2.484 |
0.115 |
0.939(0.868-1.015) |
|
Tobacco use
|
1.173 |
1.102 |
1.133 |
0.287 |
3.231(0.373-28.005) |
|
Histology
|
0.292 |
0.531 |
0.303 |
0.582 |
1.339(0.473-3.793) |
|
High level IL-10 expression in TAM
|
2.952 |
0.742 |
15.844 |
0.0001 |
19.137(4.474-81.859) |
The dependent variable is early- or late-stage group The independent variables included sex (0 = female; 1 = male), age (continuous variable, in yrs), Tobacco use (0 = Current,1 = Former,2 = Never), histology (1 = adenocarcinoma; 2 = squamous cell carcinoma;3 = others) and IL-10 expression (0 = low (<30.5); 1 = high (≥30.5). OR: odds ratio; CI: confidence interval.
Wang et al. Journal of Experimental & Clinical Cancer Research 2011 30:62 doi:10.1186/1756-9966-30-62
The increased mRNA expression of IL-10 was also associated with lymph node metastasis, lymphovascular invasion, pleural invasion and poor differentiation (p < 0.0001, p = 0.010, p = 0.017 p = 0.001, respectively).
A correlation between cathepsin B mRNA expression in TAM with NSCLC tumor T status was found (p = 0.037). Otherwise, there was no significant relationship between the mRNA expression of cathepsin B with any other clinicopathological factors (all p > 0.05).
Increased infiltration of TAMs into NSCLC correlates with a poor prognosis(5)(9). However, the mechanisms for this effect remain unclear. TAM derived molecules that function to suppress immune activation, promote extracellular matrix (ECM) remodeling may play important roles in NSCLC progression.
In the present study, the rational we selected IL-10, cathepsin B or cathepsin S, is that they were reported to be closely associated with TAMs in recent literatures(10)(11)(12)(24). IL-10 is widely known as an potent immunosuppressive cytokine associated with cancer (13)(25). It is produced by a number of cells, including tumor cells and TAMs(14)(25). Cathepsins B, cathepsin S, proteolytic enzymes, were thought to facilitate the breakdown of basement membranes thereby promoting cancer cell invasion into surrounding normal tissues. TAM expressed cathepsin B or cathepsin S in pancreatic islet, breast or prostate cancer animal models. In our study, we showed, TAM expressed high levels of IL-10, cathepsin B, but not cathepsin S in NSCLC.
Our study suggested that increased IL-10 expression of TAM in NSCLC patients correlated with late stage disease (stage II, III and IV), lymph node metastases, pleural invasion, lymphovascular invasion and poor differentiation. Although recent animal model studies indicated that cathepsin B or cathepsin S expressed by TAM play an important role in tumor progression(10)(11), and we also found cathepsin B upregulated in TAM, we failed to demonstrate any correlation between cathepsin B in TAM and stage, lymph nodal metastasis, pleural invasion or differentiation in NSCLC.
TAMs are derived from blood monocytes that are attracted to a tumor by cytokines and chemokines(14). In the tumor microenvironment, monocytes differentiate into a distinct macrophage phenotype, which is characterized by the production of high level of IL-10. TAM with high IL-10 expression level may tune inflammatory responses and adaptive Th2 immunity, exhibit anti-inflammatory and tissue remodeling functions and thereby, to favor tumor progression(14). We demonstrated that NSCLC patients with late stage disease had a higher level of IL-10 expression in TAM, which further supports this hypothesis.
IL-10 is a potent immunosuppressive factor that may promote lung cancer growth by suppressing macrophage function and enabling tumors to evade immunosurveillance(26). The potential importance of IL-10 in cancer progression is supported by reports of an association between high IL-10 levels in serum or in tumors and worse survival in lung cancer patients(15). However, other authors demonstrated that lack of IL-10 expression by the tumor was associated with a worse survival in patients with stage I NSCLC (16). The reason for these conflicting results might be that, both tumor cells and stromal(including macrophage) cells can secrete IL-10. Additionally, Wagner S et al identified that macrophage was the major source of IL-10 in gliomas(27). So it is important to isolate TAM from tumor cells to study the role of IL-10 in the progression of cancer. In our study, we demonstrated the phenotype of isolated TAM was closely associated with clinicopathological features. We can predict tumor size, lymph nodal metastasis and pleural invasion through.IL-10 expression in isolated TAM. We also found that the high expression of IL-10 in TAM was associated with poorly differentiation, which highlighted a significance role of IL-10 secreted by TAM in tumor aggressiveness.
A crucial step of cancer invasion and metastasis is the destruction of basement membrane by proteases. Recent studies showed invasion of cancer cell is increased by the proteases secreted from TAMs. Cathepsin B or cathepsin S has been implicated in the progression of various human cancers, including bladder, breast, prostate and lung cancers (17)(28)(29)(30). The cellular source of this protease in human cancers, consisting of both tumor cells and stromal cells (e.g., fibroblasts, endothelial cells, and TAMs), has remained elusive. Studies using animal models have demonstrated that TAMs are the primary source of high levels of cathepsin B or cathepsin S in prostate, pancreatic islet cancers, and mammary tumors, and its expression by TAMs plays critical roles in multiple stages of tumor growth and metastasis(10)(12)(29). Our studies demonstrated that TAM isolated from NSCLC overexpressed cathepsin B but not cathepsin S, and the cathepsin B levels were not associated with NSCLC stage, lymph metastasis, lymphovascular invasion or histological differentiation.
Interleukin-10 expression in tumor-associated macrophages correlates with disease aggressiveness of non-small cell lung cancer. We plan to conduct further studies to analyze the relationship between IL-10 in TAM and survival. The study concerning regulation of IL-10 in TAM is ongoing too. It will help to clarify and understand the possible mechanisms IL-10 secreted by TAM in the progression of NSCLC.
RW and ML designed and performed the experiment and prepared the manuscript. HQC and JZ supervised the project. YQ, SFC, XYL acquired their authorship for assistance in collecting samples and analyzing data. All authors have read and approved the final manuscript.
The authors declare that they have no competing interests.
This work is supported, in part, by National Natural Science Foundation of China (30800404), Shanghai Rising-Star Program (09QA1401200), Pujiang Talent Grant, (to J. Z), Young Investigator Grant from Shanghai Municipal Health Bureau.and Basic-clinical medicine grant (to H-Q C). We thank Shannon Wyszomierski for her editorial assistance.


